Live-Broadcasting of Bioprocesses
Real-time Monitoring of Biopharma Production
The quality specifications of biopharmaceuticals are enormous and require a long list of time-consuming analytics. A big disadvantage of these analytics so far was, that their evaluation was possible only retrospectively. The Austrian Centre of Industrial Biotechnology (ACIB) and its partners Boehringer Ingelheim RCV and Novartis Pharma have developed a new process for real-time monitoring now: integrated sensors and mathematical models give valuable information about product quality and quantity within seconds. This novel process prevents industry from product bottlenecks and allows the reduction of costs.
The market share for biotechnologically produced medicals such as vaccines or pharmaceuticals against cancer or diabetes is steadily increasing and has doubled over the last decade. The biotechnological production allows a more flexible and gentle process compared to chemical methods and opens up the synthesis of vaccines or therapeutics of diseases like rheumatoid arthritis, which have not been treatable before. Compared to chemical synthesis of medical active substances, these biotechnological processes in living cells take place in milder conditions, thus being environmentally friendly. But of course, the application of biotechnology brings along some disadvantages as well: The production in biologic systems is a challenge in terms of concomitant impurities of the product. A multilevel purification process as well as deep process understanding are essential “must-haves” for being able to meet the official purity requirements.
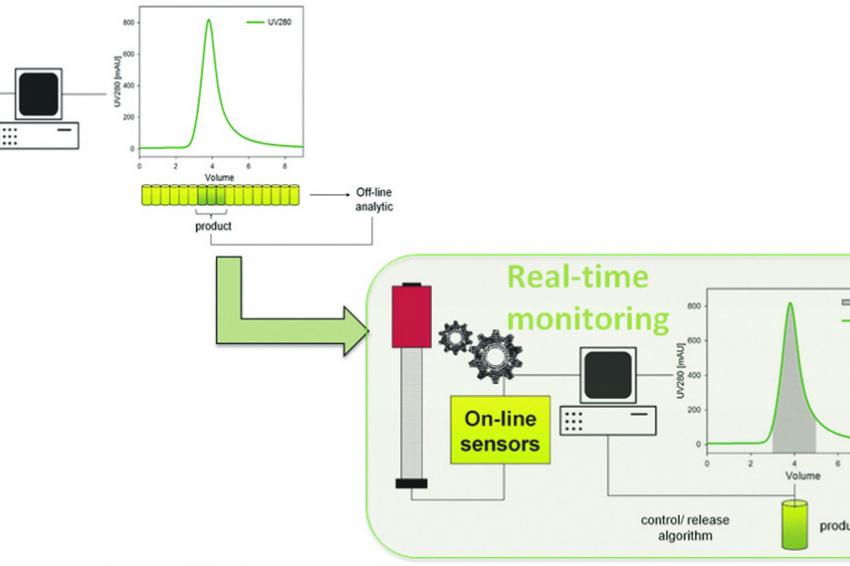
How did it work so far? Currently, quality controls are realized by regular sampling and subsequent analyses outside of the process. This comes along with huge time efforts. Apart from that, this kind of control provides information about quality and quantity only many hours after particular process steps, which means that biotechnologists are constantly lagging behind before being able to adjust process parameters for a better output. A prompt reaction is impossible. Although continuous production and quality control in real-time are state-of-the-art in fields such as automotive or nutrition — enhancing product quality and making production more efficient — these strategies found little to less attention in life sciences so far.
Real-time Monitoring through Knowledge-based Process Control
ACIB cooperated with Boehringer Ingelheim RCV and Novartis Pharma for developing a new system that enables for monitoring complex purification of biopharmaceuticals in real-time. “Previous methods gave information only about a single quality parameter in the running system. But what we urgently need, is a monitoring process that gives information about product quality and quantity as well as about impurities within seconds. This is exactly what we achieved with our new method: We combined different sensors and mathematical models that connect measured signal data with important quality criteria. The keyword is knowledge-based process control”, explains Astrid Dürauer, project leader and key researcher at ACIB as well as senior scientist at the University of Natural Resources and Life Sciences (BOKU) in Vienna, Austria.
The research group developed a user interface that integrates the monitoring of the system, the visualization of measured data as well as the transfer of information and direct response of the control unit. Dürauer is more than happy with the new process: “This system significantly reduces the risk of manufacturing errors, the total process duration and the extent for analytics. Industry benefits from a safer, faster and more efficient process.” A side effect of increased efficiency is the new availability of production capacities. Manufacturers can produce more different products or more batches of one product. On the long-term, manufacturing costs are reduced, and resources are saved.
Application in an Industrial Environment
ACIB developed the prototype at Boehringer Ingelheim’s Vienna site together with both industrial partners and tested it over a period of more than 3 years. Quite recently, the system was successfully put into operation by both company partners and a patent for Europe as well as the USA is in place. For the manufacturers the system means a quantum leap in production, as Christian Eckermann, head of Biopharmacy at Boehringer Ingelheim RCV states: “From a technological point of view the in-process quality control approach in real-time is a mandatory condition for a real-time approval of produced batches, which further guarantees an efficient production of high-quality biopharmaceuticals. The system is an important basis for reduced process duration and offers a significant innovative advantage in terms of process control during the production of biopharmaceuticals!”
This is confirmed also by Michael Kocher, country president of Novartis Austria. In his view, the technology is an important reaction to the governmental demand for process control in the pharmaceutical industry. In other branches such as automotive or food industry continuous production and quality control for real-time production are state of the art and have shown a significant improvement in product quality as well as higher production efficiency. According to relevant authorities it is high time for the implementation of real-time controls also in pharmaceutical industries for drug approvals. However, the biopharma sector was lacking appropriate strategies for successful implementation. Kocher states: “The new system has high potential to herald biotechnological production into a new era!”.
Contact
ACIB Austrian Centre of Industrial Biotechnology
Petersgasse 14
8010 Graz
Austria