Economic Impact and Challenges of Continuous Pharma Processes
Optimized Production of Active Ingredients and Finished Drugs
The strong focus on supply chain security will increase cost of pharmaceutical production in years to come. In this context, companies considering production in mid to high labor cost countries need to carefully analyze their conversion cost development. It is precisely in this situation that optimized production of synthetically and biologically produced active ingredients and finished drugs is extremely important.
Compared to the incremental optimization of existing processes, the automation of individual production steps up to complete continuous production offers significantly higher efficiency potentials.
Continuous Production Approach
Continuous production links individual production steps together and quality-relevant parameters are monitored in real time by so-called in-process controls. In the ideal case, the entire production process can be carried out continuously, from the weighing of the sample to the finished drug. In this case, production is controlled by predefined parameters, compliance with which guarantees the quality of the end-product.
Regulatory authorities in several countries support the introduction of continuous production, as the continuous monitoring of relevant parameters guarantees a higher product quality than the random checks used in the classic batch process. In addition to the higher product quality, continuous production offers five economic advantages for the pharmaceutical industry, which will be discussed below.
1. Smaller Production Facilities
In classical manufacturing facilities, the production process is carried out in a stepwise approach. Intermediate products are stored in containers which take up a lot of space, until they are released by the quality department and the process continues. In continuous production, these containers are not needed, as individual production steps are linked together by conveyor systems. Furthermore, continuous production allows the use of smaller equipment for the same production quantities. In consequence, smaller machines and the continuous process are less labor intensive and enable a more efficient production.
2. Increased Equipment Efficiency
Changeover between individual production campaigns has become complex in recent years due to new regulations like the cross-contamination guideline. In addition to the costs for the preparation of these cleaning protocols, the cleaning times reduce the efficiency of the machines used, in some cases considerably. It is not uncommon that the cleaning of the machines used takes longer than the production campaign itself. Continuous production equipment is frequently easier to clean or even dedicated for a specific product and can help reduce this requirement and increase efficiency.
3. Faster Product Release
Real-time monitoring of quality-relevant parameters eliminates the need for quality checks of intermediate products during continuous production. The process is not interrupted when waiting for individual releases from the quality department and there is no need for storage of intermediates. The final release is also much faster and completed to a large extent, by adhering to the described process parameters. In addition to the pure time savings, this helps to reduce the number of quality tests and increases the efficiency in the quality control department.
4. Lower Energy Consumption
Reducing the size of production sites usually has a positive impact on energy consumption, especially for heating, ventilation and air conditioning.
Furthermore, continuous production allows the product to be completely shielded from environmental influences during the production process. This shielding allows, for example, the production of sterile products in clean room classes with lower requirements for air exchange and other parameters. The lower requirements help to reduce energy consumption.
5. Reduced Waste
The elimination of upscaling during product development as well as the use of smaller machines in the production process can help to reduce waste of pharmaceutical intermediates and end products. Furthermore, real-time monitoring of critical parameters allows to react promptly to deviations by adjusting the production process. This can help to prevent the destruction of entire batches in the event of a failed final release.
Summary of Positive Economic Effects
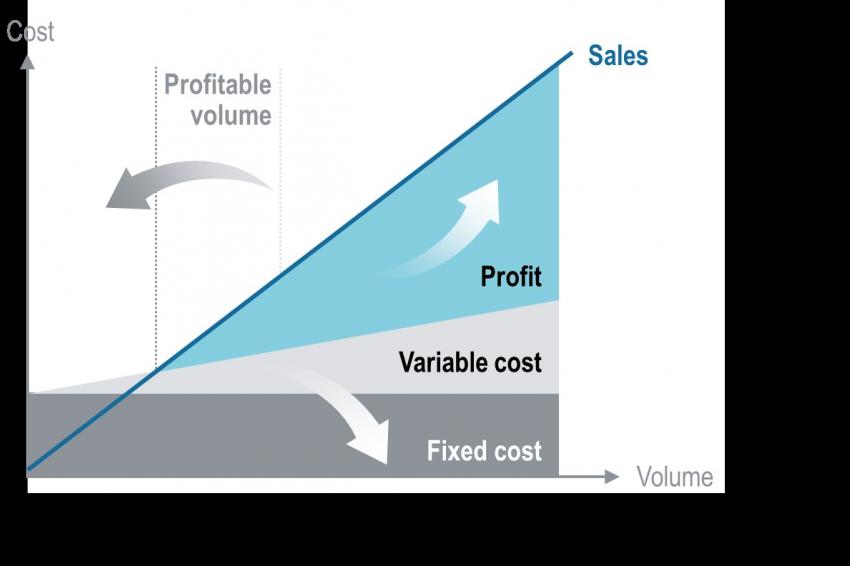
The reduction of operational costs (up to 30%), the higher operational efficiency (up to 80%), the reduced lead times (up to 90%) and the decreased product deviations (up to 50%) contribute to a reduction of variable costs. In summary, this makes production of boarder line products possible and improves profitability of existing portfolio products (Fig. 2). Especially, the labor cost reduction (up to 50%) contributes to the potential reallocation of pharmaceutical production and can help to secure supply chains.
Limitations of Continuous Production
However, from a technological point of view not all processes can be converted into a continuous process. The process development is usually still very costly and takes considerable time in the development phase. Therefore, continuous production is mainly recommended for launch products and few existing portfolio products. For these reasons, pharmaceutical manufacturers still rely primarily on classic batch production despite the identified savings potential.
Business Case of Continuous Production
The exemplary calculation of savings potentials per category for an exemplary transfer from a classical batch production into a continuous production process demonstrates a cost of goods sold (COGS) saving potential of ca. 30%. Main contributors are the lower facility costs (ca. 25%) associated to the described smaller production facility and the reduced labor costs (ca. 50%) due to automation and continuous production. Additionally, savings on raw material and consumable consumption (ca. 25%) help to achieve the described COGS saving potential. Saved COGS directly influence the product‘s profitability and demonstrate the huge potential of continuous production processes.
The Drivers
For equipment suppliers the adoption guarantees new equipment sales, besides that machine manufacturers are extremely interested in obtaining data from production processes. As service providers, the machine manufacturers are establishing a second business segment supporting the pharmaceutical companies in the analysis of their process data. From our perspective, machine manufacturers are ideally positioned for this, as they are both familiar with the technical possibilities of their equipment and can collect data from all production sites using their equipment. Especially in times of increasing competition from low-wage countries, which are now also supplying high-quality machines, the collection, analysis and processing of data is crucial for the future success of machine manufacturers.
Summary
Innovative pharmaceutical companies in particular have a strong focus on delivery reliability of their products from an ethical and economic perspective. Reliable supply chains are often guaranteed by expensive safeguarding mechanisms. Stable and flexible continuous production processes can help to reduce these security mechanisms and thus save costs.
Continuous production fits perfectly into an increasingly data-driven value chain. The collection, analysis and evaluation of data is becoming more and more important and contributes to corporate success. Especially in personalized medicine, as can be seen in the example of recently launched CAR-T
cell therapies, better control of one‘s own supply chain and the offer of digital solutions for coordinating patient-specific production are already decisive for the commercial success or failure of companies.
For these reasons, classical pharmaceutical companies should increasingly rely on digitally networked and automated production in order to optimize the costs of classical products as well as to lay the foundation for the introduction of innovative products today.