Managing Supply for Complex Biotherapeutics
Drug Substance Manufacturing of Antibody-Drug Conjugates (ADCs) Requires an Experienced CDMO
Antibody-drug conjugates (ADCs) are complex bioconjugates typically used as vector-based chemotherapy, allowing the selective delivery of a nanomolar potent cytotoxic agent within a tumor. For drug substance manufacturing, the cytotoxic agent and the targeted monoclonal antibody are chemically linked via a linker. The drug product for an ADC is typically filled and lyophilized to distribute the drug in a stable form.
ADCs are complex, and their manufacturing supply chain contains five different technologies with specific asset needs: monoclonal antibody, cytotoxic payload, linker, bioconjugation and fill & finish. Even with the necessary expertise, the combination of disparate technology requirements to manufacture the five ADC components creates supply complexity. It can take years of effort to establish a reliable, high-performing logistics network to manufacture ADCs. Many companies, therefore, consider outsourcing to experienced contract development and manufacturing organizations (CDMOs) as an option to accelerate development and limit risk.
The Global ADC Portfolio Continues to Grow
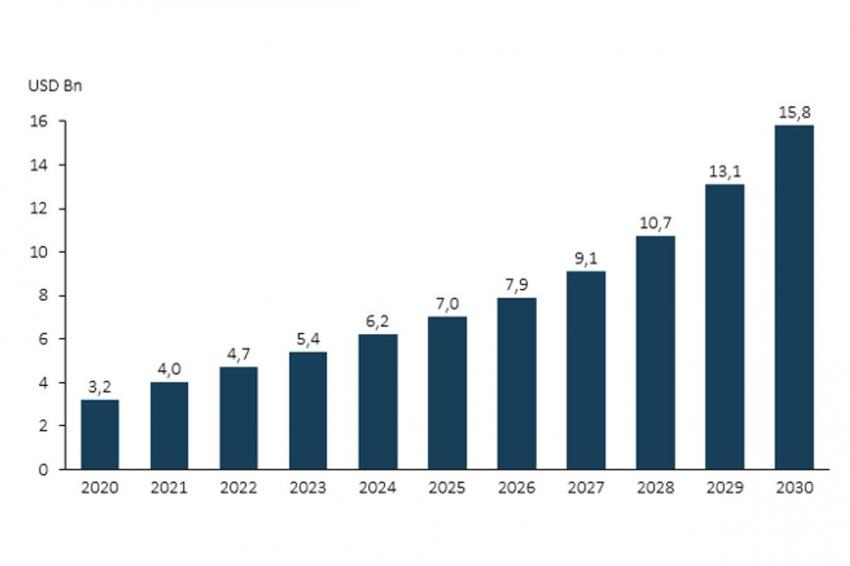
Antibody-drug conjugates have shown an exciting pace of development, especially over the last 10 years. Today, we have 10 approved ADCs and more than 80 candidates in different clinical studies. There are substantial learnings from the successful candidates, but also from over 50 candidates that were abandoned in the clinic over the same time for reasons of toxicology and efficiency. Advancing technology has improved and will further improve the probability of success by better allowing control of toxicity and a wider therapeutic index. According to Lonza‘s market data analysis, ADC sales will rise to over $15 billion over the next decade.
Lifecycle and Supply Considerations
Successfully managing the supply of the various components of ADCs requires a sophisticated understanding of the technological development process and access to a range of large- and small-molecule manufacturing assets.
In the early stages of development for monoclonal antibodies, it is important to create processes that are adaptable and resilient through later stages. Proper control of quality attributes with analytical methods ready for the purpose must also be established. The use of established expression systems and platforms substantially reduces risk and the time needed to submit an Investigational New Drug (IND) application. Small molecule assets are required to deliver the linker and cytotoxic payload. While the linker can typically be manufactured in an asset without special highly potent capabilities, the cytotoxic payload requires special safety control. Payloads for ADCs come with an inhibitory concentration (IC50) which is typically below picomolar concentrations.
This toxicity is translated into occupational exposure levels, which are typically below 10 ng/m3, requiring proper strategies for substance handling during manufacturing. Substantial resources are required to invest in cytotoxic-dedicated infrastructure, and define and implement processes and control strategies. Proper training and control measures are continuous efforts through development. Beyond infrastructure and established safety concepts, the supply of cytotoxic payloads requires development and technology transfer capabilities that support process implementation at the required scale. Because the effort to execute activities for cytotoxics is high, good planning and effective execution are key.
Regarding bioconjugation, the handling of cytotoxics in a bioburden-controlled environment requires special attention from a safety and quality perspective. Specialized biologics assets must be equipped to fulfill these requirements. Bringing the two worlds of small molecules (payload) and biologics (antibody) together in bioconjugation typically requires a dedicated workforce specially trained in this expertise. Process definition, implementation and control are critical since high-value intermediates need to be conjugated. The bioconjugate is isolated in the final and stable liquid formulation as bulk material is stored frozen for control. Storage and shipment processes need to be in place to prevent any unplanned supply interruption. The conjugation can be made in different ways and with the high number of bioconjugation technologies developed over the recent years, the early development of lead candidates needs special attention.
For the fill and finish of ADCs, assets need to be equipped to handle cytotoxic drug formulations. Since formulation and processes need to be developed and implemented, companies delivering drug product need to have the necessary infrastructure and knowledge to cover process development and manufacturing. For product testing and stability testing, there is a significant overlap with drug substance. Depending on the level of integration of the offering, synergies can sometimes be realized to streamline the effort.
Simplified Supply Chain with the Right Partner
Proper management of the complex ADC supply chain is an important success factor in making these life-saving drugs accessible for patients. Since the supply of monoclonal antibodies, cytotoxic drugs, bioconjugation and fill & finish require expensive assets and expertise, small biotech companies typically do not have the required in-house capacity. At the same time, established pharma companies understand the advantage of outsourcing to secure a reliable and efficient supply.
Bringing the entire lifecycle together under one roof simplifies the supply chain substantially. This integration removes complexity from an overall chemical manufacturing control (CMC) management. A high level of integration allows for easier planning, contracting, quality systems, program management, analytics, storage, shipment and improved timelines.
Currently, there are few CDMOs that have invested in capabilities across all the technologies required for ADCs. Fewer still have mastered manufacturing processes at the level
needed for reliable production. Lonza entered the ADC market in 2006 and is today the most advanced supplier to provide an integrated supply for ADCs under one roof. The fact that Lonza is part of the supply chain for the majority of the 10 commercial ADCs, having completed over 750 successful cGMP batches, illustrates the importance of outsourcing and experience for this product class.
The Future of ADCs
The dynamic market outlook for ADCs is very positive. With ten commercial ADCs in production and a healthy clinical pipeline in place, sales are projected to surpass $15 billion in the coming years. ADCs are widely approved for the treatment of solid tumors and will offer an answer to many unmet medical needs outside oncology as non-cytotoxic bioconjugates are advanced. However, bioconjugates currently have one of the most complex supply chains in the pharmaceutical industry. By outsourcing to a reliable CDMO, pharma & biotech innovators may access the expertise, infrastructure and flexible business models needed to secure supply. An experienced and reliable CDMO partner can provide access to supply under one roof and help deliver new, innovative drugs to market and to patients sooner.
References for this article can be requested from the author.
Iwan Bertholjotti, Director Commercial Development Bioconjugates, Lonza, Visp, Switzerland