Supplier Controls in the Healthcare Industry
Clariant and LyondellBasell Respond to Challenge
Poor Controls - In the past five years, several contaminated products have led to patient and end-user injuries and death globally. In 2007, there was diethylene-glycol contaminated toothpaste, and millions of toys were recalled due to lead contamination. In 2008, contaminated heparin was recalled by the U.S. Food and Drug Administration (FDA) and melamine-contaminated milk caused thousands of adverse events and several infant deaths. Investigations into the causes of these adverse events found that, among other reasons, inadequate or poor supplier controls were a factor.
These incidents have triggered U.S. and European regulatory bodies to increase their vigilance on supplier controls for the medical device and pharmaceutical industries. In December 2008, the Global Harmonization Task Force (GHTF) issued a guidance document that outlines the steps medical device manufacturers should include in their supplier control process. A second document issued in November 2009 (in draft form) describes the techniques for inspecting a medical device manufacturer's supplier controls.
The level and extent of controls should be based on the criticality and risk of the products or services that could affect the safety, effectiveness and quality of the final product. It is the responsibility of the manufacturers of the finished product or device to determine and communicate those requirements to their suppliers. The benefits of good supplier controls translates to effective quality management systems and risk management processes, high quality and consistent products, lower scrap, improved productivity, and fewer adverse events, complaints and recalls.
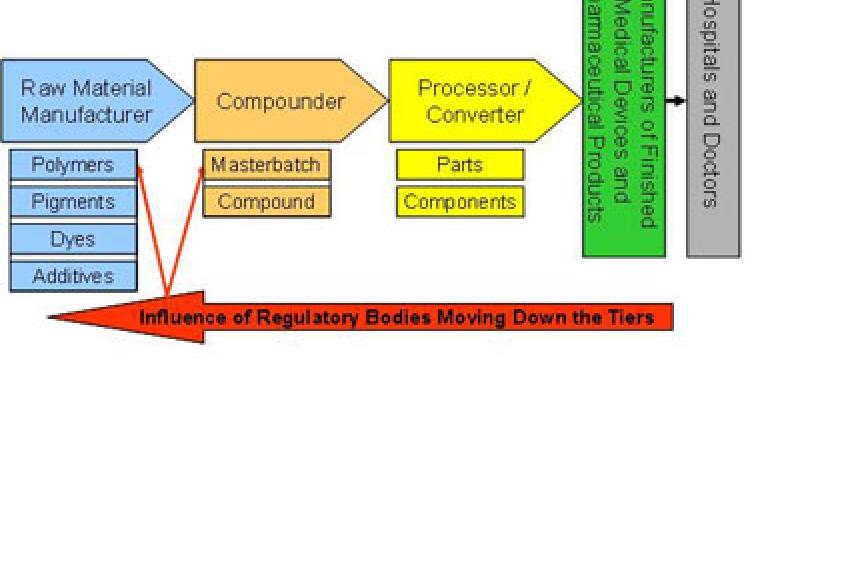
More Rigor And Control
Component, part and material suppliers to medical device manufacturers and the healthcare industry must be prepared to expect much more rigor and control from their customers. The level and extent of controls on suppliers will not only depend on the criticality and risk of the product they supply, but also on their position in the supply chain. Tier 1 suppliers will see more controls versus Tier 2 and Tier 3 suppliers (fig. 1). Wherever material suppliers fall in the value chain, they must still ensure that their products meet all the performance and regulatory requirements needed for the specific application.
Understanding The Risks
Many companies in the supply chain do not fully understand where risks and changes to their products can come from. In some cases they see the medical device and pharmaceutical market as high risk and of low business interest. A few suppliers have developed a dedicated approach to supplying their products and services to this market. Clariant and LyondellBasell work closely with each part of their supply chains to mitigate risks posed not only to themselves and their direct customers, but ultimately to the pharmaceutical and medical device companies.
Clariant is a supplier of masterbatches and compounds, and LyondellBasell is a supplier of polyolefin resins to a diverse industrial base including the healthcare industry. Each company has served the industry for decades, and has adopted similar approaches to ensure that their business processes and healthcare products are well positioned to meet the challenges associated with more stringent controls.
The medical device and pharmaceutical industries continue to use plastics with high-performance properties and aesthetics in ever-increasing amounts. These materials must meet specific properties such as sterilization resistance, chemical and lipid resistance, and biocompatibility. Slight differences in impurities, extractables or leachables could potentially affect the biocompatibility and toxicity of the final formulated product.
The U.S. Pharmacopoeia (USP) and the European Pharmacopoeia (Eur.Ph) require detailed information on the material components and formulation, manufacturing processes, and extensive supporting data with respect to physical and mechanical properties, chemical resistance, biocompatibility and toxicity tests. The results must be maintained in specific files, or the material must meet required properties set in standards.
The advantage of this system is that manufacturers of finished products could use these materials in their designs and products with the confidence that they would meet regulatory and application requirements. The disadvantage however, is that this file or record is a point-of-time submission and is material-specific and formulation-specific. Managing any material or formulation change over time is thus not easy and becomes a formidable task both from regulatory and business perspectives.
Clariant And LyondellBasell
In order to meet the ever changing requirements and needs of both the healthcare industry and their customers, Clariant and LyondellBasell have implemented strategic business processes that involve working with their customers (developers and manufacturers of finished healthcare products, converters and processors, trade associations etc.) and their suppliers (resins, pigments, additives, catalysts etc.) to manage, control and mitigate the risks associated with the products they supply to the healthcare industry.
Clariant has several business units (BU) that supply the medical device and pharmaceutical industry with products such as intermediates, excipients and masterbatches. BU Masterbatches is a leading player in color and additive concentrates and compounds for the plastics processing industry across diverse applications from consumer electronics, toys, food, personal care packaging, textiles and automotive parts.
The business unit is concentrating the know-how at three dedicated centers of competence across the globe with offering the possibility of complete manufacturing line-segregation capabilities for medical grade materials. These centers have a global quality management system that is based on current good manufacturing practices and that uses the ISO 13485 standard for medical devices.
Processes and procedures instituted for medical products include fully traceable production lots, an "open to audit" policy (a policy that encourages both direct customers and the brand owner to carry out audits) and rigorous change control processes. The result has been introduction of two new internal classifications of manufacturing for products used in medical and pharmaceutical applications referred to as MedCat 1 and MedCat 2 and the introduction of a new brand to distinguish products and services from the conventional products.
The levels of controls with respect to material change notification, process controls, risk management, quality management systems and documentation, are different from the normal processes used in standard compound and masterbatch production sites.
LyondellBasell's Purell is a brand of polypropylene and polyethylene developed for use in the healthcare industry. The value of Purell products includes consistency of formulation, continuity of supply, single sourcing, backup plants and pharmacopoeia compliance. LyondellBasell was the first polyolefin company to launch a dedicated group of products used in healthcare applications, with dedicated internal quality systems and procedures that exemplify the spirit of pharmaceutical and medical device good manufacturing practices. Risk management procedures are also an integral part of this process.
Using Risk Management
Clariant and LyondellBasell work cooperatively to educate medical and pharmaceutical suppliers. They are building awareness of the importance and significance of change controls as they are applied to polymers, masterbatches and compounds. Using risk management to control products used in the healthcare industry is also part of their strategy. Both companies recognize that risk increases with the complexity of the supply chain, and have developed strategies to mitigate such risks and manage raw material changes, consistency and quality.
Clariant has been working with their suppliers to provide pigments and additives with consistent quality and purity. The less the variation in these chemicals, the better the confidence that the masterbatches will not change their biocompatibility and toxicity properties from lot to lot or batch to batch.
CoA Based on Macroscopic Properties
The macroscopic properties of polymers and resins such as viscosity, flow, and physical and mechanical characteristics are well understood, and can be controlled and measured. These physical properties are used in the evaluation and selection of materials used by customers in healthcare applications.
Certificates of analysis (CoA) and quality metrics are also based on these macroscopic properties. LyondellBasell recognizes that properties specific and critical for healthcare applications such as leachables and extractables, biocompatibility and toxicity are highly dependent on the resin's microscopic properties such as impurities, molecular weight distribution and low molecular weight oligomers.
The Importance of Supplier Controls
Supplier controls are becoming an important aspect of the quality systems for finished product manufacturers in the healthcare industry. Inspections by regulatory bodies will be placing an increased emphasis on purchasing controls to ensure that these manufacturers exert the appropriate extent and level of controls on their suppliers based on product risk and criticality.
Clariant and LyondellBasell have taken a proactive approach to building awareness among their customers and suppliers of the importance of mitigating risk, minimizing change and maintaining compliance and consistency of their products supplied to the healthcare industry. In so doing, these companies are well-positioned to serve customers who require preferred suppliers that have understood and implemented risk-based quality systems for the healthcare industry.
Contact
EMG The European Marketing Group BV
Lelyweg 6
4612 PS Bergen op Zoom
+31 164 317025
+31 164 317039