Serialization – the Bigger Picture
Preparing for All Requirements with a Scalable Machine and Software Concept
Potency pills or even life-saving pharmaceuticals such as heparin – the trade with counterfeit medicine is still booming. Accordingly, drug producers and contract packers are shifting their focus to serialization of pharmaceutical packaging. Time is pressing, as numerous laws and guidelines have already or will come into force all over the world in the coming years. Companies concerned are facing the challenge of finding and implementing appropriate technologies, and connecting them with their manufacturing and packaging processes. A scalable machine and software concept undoubtedly is the safest option.
The European Union‘s Falsified Medicines Directive 2011/62/EU came into effect in February 2016. It stipulates the usage of coded packaging with unique serial numbers for all prescription drugs. Similar to many other countries, the serialization feature is a 2D data matrix code. It contains a randomized serial number, the batch number and expiry date, as well as further data, if required. At the same time, the EU demands a second level of security in form of tamper-evident closures, such as integrity seals or glue, which are recorded in the CEN norm DIN EN 16679:2015-03. They clearly indicate whether a package has been previously opened or tampered with.
Toward Complete Connectivity
On November 27, 2013, the Drug Quality and Security Act (DQSA) was adopted in the U.S. In early 2015, the implementation began on batch level. As of 2017, serialization of primary packaging will be mandatory. The biggest challenge for all parties involved, however, will follow in 2019, when the serial number of each individual pack and outer packaging must be known to pharma wholesalers – either in form of the National Drug Code (NDC) or the Global Trade Identification Number (GTIN). First, all drug producers must comply with this law. In an interval of one year each, repackers, wholesalers and dispensers will follow. Exactly ten years after commencement of DQSA, that is end of November 2023, the complete connection and aggregation is supposed to be implemented. Based on the unique serial number, products can then be electronically traced on all aggregation levels, as well as on batch and single packaging level along the entire supply chain through to the dispensing point – unambiguously and in real time.
In parallel, many other countries are developing and implementing new guidance and laws. In the MENA region (Middle East and Northern Africa), efforts for drug traceability are already underway. Saudi Arabia, for instance, initiated the first phase in March 2015 with obligatory data matrix codes on pharmaceutical packaging. Phase 2 – actual serialization – will come into force in 2017. While phase 1 only requires a machine to print data matrix codes, phase 2 necessitates producers and packers to have machines for serialization as well as an IT infrastructure to generate the serial numbers. Moreover, they must be able to send the numbers to their machines, report the results to manufacturers‘ databases and record them.
Significant Differences
Now what are the fundamental differences between the different implementation stages? On the one hand, the EU attributes special importance to tamper-evidence. On the other hand, the U.S. wants to implement the first aggregation step at a very early point in time, thus changing the requirements for both local pharma companies and importers. While serialization is currently still often seen as regional responsibility, the different legislations will have a global impact. Many large drug producers operate production facilities in different countries and export their products all over the world. Equipping packaging lines according to only one standard would be very short-sighted.
Take a practical example: a large generic producer from the MENA region sells his products in the local market, as well as in Europe and the U.S. The majority of pharmaceuticals are packaged at the production sites in Jordan and Saudi Arabia. Against this background, all pharmaceutical packaging must conform to different national serialization standards. Accordingly, flexible solutions which are able to fulfill far more than just one national guideline are highly sought-after. The same accounts for contract packers, who must comply with the laws of the respective market, as well as with the requirements of their clients.
Adapting Established Processes
In general, most producing and packaging companies will not be able to avoid developing and applying a global strategy to consistently assign serial numbers. This requires solutions that are compatible with in-house processes, leading to the adaptation of often highly demanding packaging processes. Furthermore, new processes must be established for both management and storage of serial numbers. The requirements can be subsumed in a single key word: “modular”. A comprehensive serialization solution should not only be able to mass-serialize the packed product, verify the codes and apply labels or tamper-evident seals to the packaging. The entire process needs to be controlled, and the data retrieved at any time. Only this way do producers – and later on the dispensing points – have an exact overview of all process steps.
To this end, Bosch Packaging Technology has developed the CPI software for Track & Trace, which can be easily integrated into existing IT infrastructures. The company can rely on the own experience from the automotive sector, where automation and connectivity of machines, processes and IT have been tried and tested for years. Logic and functionalities have been transferred and adapted to the specific pharmaceutical requirements for Track & Trace applications.
From Machine to Database
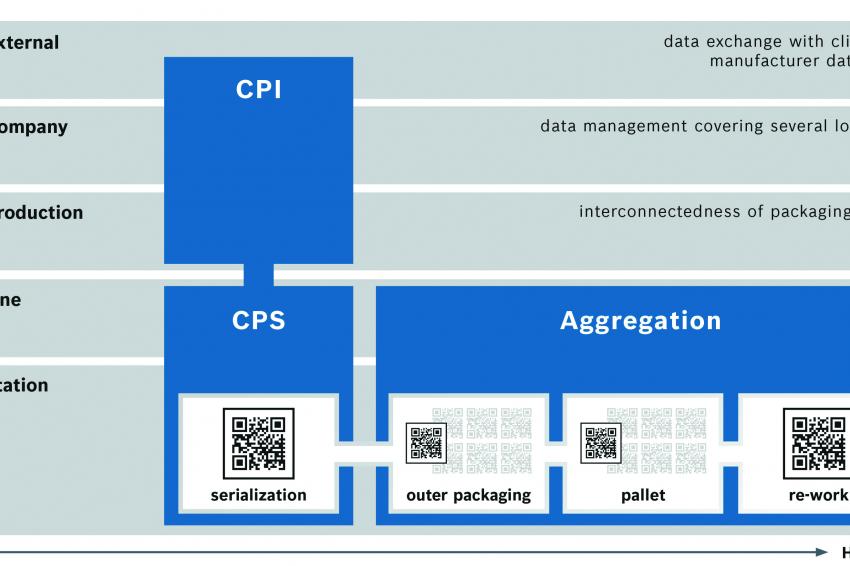
How can we imagine a holistic serialization process in reality? It starts on the application level (level 1). Here, the modular CPS system from Bosch serializes up to 400 folded cartons per minute. The camera system automatically verifies the printed tracking data. Subsequently, a security seal is applied by the Tamper Evident labeler. The modules can be operated via a central user interface, and the data can be documented (level 2).
To control both operating condition and data at any time, the connection between the physical machine level and the control software must be integrated across many stages of the company’s IT (level 3). From this level onwards, the CPI solution for Track & Trace ensures that the entire production environment or several sites are depicted (level 4). Moreover, an interface is also possible for data transfer, for instance to the SecurPharm database or to the customer (level 5).
The Highest Flexibility
Back to the example of the generic producer, who has lately equipped numerous packaging lines with CPS modules and CPI software. The company benefits especially from the new data connection between all lines that are situated at different locations and can now be monitored from the headquarter. Thus all relevant data is available in the entire company network, and all results are recorded in the audit trail. The data sets are bundled with the production results according to aggregation specifications, and sent back to the CPI software. From here, they can be transferred to regulatory or producer-owned databases.
Depending on country and guideline, the serial numbers are either allocated centrally or generated by the company. Contract manufacturers in turn receive the numbers from their clients. This requires very high flexibility within the process cycle. The CPI solution from Bosch is equipped for all three cases. Companies can not only manage serialization from the allocation of the serial number through to the last aggregation step. They can also flexibly connect single components, third party machines, packaging lines, own or third party IT systems and entire factories with each other according to the respective guidelines. Hence it really is worth while looking at the bigger picture – for manufacturers and contract packers, as well as for mechanical engineering companies with a focus on international customers.