Quality and Efficiency in Pharma Manufacturing


The pharmaceutical industry is confronting a variety of new and complex challenges in the face of globalization and changing market dynamics. The past decade has witnessed a decline in the discovery, approval and marketing of new chemical entities with fewer blockbuster drugs making it to the market, as well as stiffer competition from generics. Stricter regulatory requirements demand continuous investment to track products and document processes.
The pharma industry stands out among manufacturing industries for the astronomical amounts it spends every year on research and development - a stark contrast to the relatively low-tech level of manufacturing.
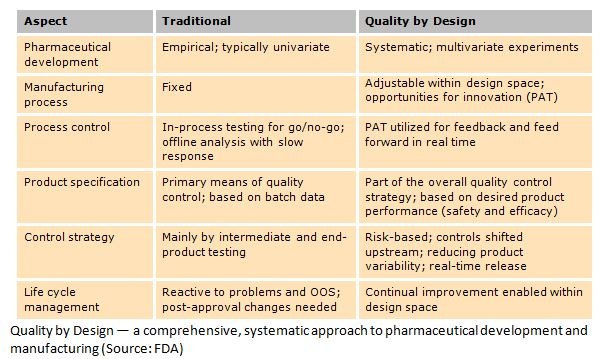
Pharmaceutical manufacturers are addressing the challenges of manufacturing efficiency with stronger cost-containment measures and initiatives to improve operational excellence and quality. In North America, the U.S. Food and Drug Administration (FDA) has championed one such initiative, known as Quality by Design (QbD), with the goal of improving productivity and reducing manufacturing costs for U.S. producers. This initiative is part of a larger effort to reduce overall health-care costs based on the assumption that drug costs are high because of inefficient manufacturing.
Process analytical technology (PAT) is an enabling technology for QbD. PAT tools enable in-line measurement of critical quality attributes and process parameters in real time, allowing running processes to be adjusted on the fly. Product quality is verified in multiple steps during process execution rather than performing QC at the end of the line, helping to shorten batch-release times. In addition, correcting the course of a batch that is threatening to go out of spec can dramatically improve yield. The business benefits of PAT tools are promising: They include more efficient use of equipment, higher batch yields, less waste, shorter time-to-market, and significantly better process understanding and control.
The Problem of Quality by Testing
Quality by testing is the traditional approach to quality control that compares finished drug products to an approved specification and rejects anything that doesn't meet it. Quality is assured by testing materials before and after manufacturing, all within a strict, fixed process. This process must be tightly controlled because of the uncertainty of whether the specification alone is adequate to ensure quality. The downside is that overly stringent specifications may result in unnecessary recalls or drug shortages.
The inherent problem in this approach is that root causes of failure are poorly understood, and as a result, manufacturers risk ongoing, unexplained losses. This can continue indefinitely or until supplements are filed with a government authority to allow broader acceptance criteria. But changing validated processes is prohibitively expensive, so quality is maintained through rigid manufacturing practices and by testing end products, without considering how system and process design can ensure product quality.
This method also creates problems during the move from laboratory pilot to full-scale production. Production specifications are typically derived from test data from small batches in the laboratory environment. Scaling up to commercial production can reveal new complexities that were not evident during development. As production levels are reached, the regulatory requirement of filing supplements for minor and incremental changes to manufacturing processes makes it difficult to improve the process with closed loop feedback to achieve continuous, real-time quality assurance.
Quality by Design
One of the most talked about movements in QA in recent decades is Quality by Design (QbD), a methodology that assumes quality can be designed into a product. According to QbD, most quality problems are a result of the way quality was planned (or not planned) in the first place. Instead of raw material and end-of-line testing, QbD focuses on designing and developing manufacturing processes to ensure that predefined product quality requirements are met. This built-in quality originates with the careful design and control of these processes. The result is a well-understood product that consistently meets quality specs.
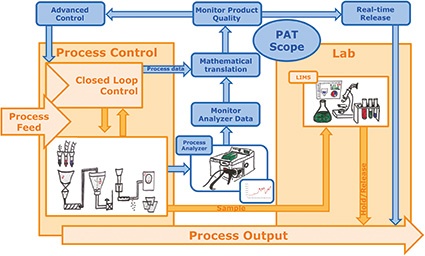
PAT refers to the measurement and evaluation tools that enable QbD. PAT tools consist of in-line sensors and analyzers (spectroscopy instruments, e.g., NIR or near-infrared) that measure substance properties during the manufacturing process and pass on these data in real time for evaluation. Software tools translate these data into product quality information (the CQAs) that infer final quality of the batch and can be used to adjust the running process to ensure that targeted quality goals are achieved. Tools for advanced process control (APC) play a role here.
In support of QbD, PAT's goal is to ensure final product quality by designing, analyzing and controlling process variability through timely measurements of critical quality and performance attributes of raw and in-process materials. This applies not only to manufacturing processes, but also to development. With in-line analytics, product developers can gain a much higher and faster understanding of the process than is possible with offline analysis (manual sampling). The real-time access to information offered by PAT is invaluable during pharmaceutical development and during the scale-up to commercial production.
PAT In Product Development
Within the QbD framework, PAT tools can be applied as early as the development phase of a new product. Once an active ingredient has been developed in the laboratory, the next step is to decide on dosage and delivery (tablet, capsule, suppository), and which excipients to use. These factors then determine the processes used to manufacture the drug product.
During development, in-line analytics allow faster and clearer understanding of process dynamics versus traditional offline analysis. Performance requirements at this stage will differ significantly from those in the later manufacturing process, but the parallel application of these tools in both areas allows a better understanding of the specific challenges of the process. It also facilitates scale-up to production levels thanks to a deeper process understanding.
PAT in Process Design
PAT is also an integral part of manufacturing process design. The use of PAT tools enables users to develop robust, well-understood processes with defined parameters that allow for real-time monitoring and adjustment of CQAs. During process development, raw materials, process parameters and CQAs are investigated to determine raw material attributes, process parameters and CQAs for each process, and to identify relationships among them. In the scale-up to commercial manufacturing, parallels to the development phase can be exploited to shorten process development and time-to-market. Knowledge is not static - rather it builds throughout the manufacturing life cycle, and the deployment of PAT tools along this cycle supports this notion.
Real-Time Release Testing
In the pharmaceutical industry, strict regulations and regular quality checks can lead to frequent, costly delays and unnecessary scrapping of materials. This creates inefficiencies that lower overall productivity. The idea of measuring product quality in real time with in-line analyzers has been talked about for a decade, but implementation is still relatively new, despite compelling business benefits.
Real-time release testing (RTRT) is defined as the ability to ensure product quality during the process based on real-time process data instead of end-of-batch QC. RTRT uses PAT tools to gather data from analyzers and other inline process measurements to generate quality information that can be used to adjust the process during batch execution. While it doesn't replace the review and quality control steps required by good manufacturing practice for batch release, RTRT can eliminate end-product quality testing, cutting out delays and streamlining the whole manufacturing process. A side benefit is that the integrated quality information automatically becomes part of the batch record.
PAT in Continuous Processes
Manufacturers often employ batch-control methods with batch sizes determined by production schedules, capacity, or specific customer orders. While continuous processing is known to have distinct advantages over batch, quality-control requirements force pharma manufacturers to think in terms of finite lot sizes. However, that mentality is changing - thanks in part to the availability of PAT tools.
By providing continuous, real-time quality information, PAT essentially turns a batch process into a continuous process, allowing constant quality checks along the way that eliminate the need for a final quality check to release a batch. Instead of producing finite quantities, manufacturers can "stream in" and "stream out" enough raw materials and finished products to meet demand. The advantages of a continuous process lie in the more efficient use of equipment, higher productivity and better quality.
Quality by Design takes a different approach to ensure consistent levels of quality by allowing for flexibility during the manufacturing process. PAT tools play a crucial role in the implementation of QbD by providing the real-time feedback that allows operators to keep CQAs in spec by adjusting processes during operation. At the same time, quality data collected during the process can take the place of end-of-line QC, drastically cutting batch release times. The many business benefits of employing PAT tools are highly attractive.
Company
ARC Advisory Group GmbH & Co KGStadttor 1
40219 Düsseldorf
Germany
most read

Specialty Chemicals in a Shifting World
Jennifer Abril, President & CEO of SOCMA, discusses the impact of new tariffs and the importance of regional supply networks in the specialty chemical industry.

Rugged Tablets: How to Successfully Digitize Hazardous Areas
Digital processes in hazardous areas? Durabook's rugged tablets are ATEX-certified for industrial use.
ISPE Good Practice Guide: Validation 4.0
The Validation 4.0 Guide provides a comprehensive approach to ensuring product quality and patient safety throughout a pharmaceutical product's lifecycle.

Substances of Concern
The EU Chemicals Strategy for Sustainability (CSS) highlights the shift to a hazard-centric approach in EU chemical regulation, emphasizing 'Substance of Concern' over risk-based measures.

“Access to Talent is a Crucial Factor”
In an interview with CHEManager, Edgardo Hernandez, President of Lilly Manufacturing, explains the strategy behind the ambitious investment project.




