Quality and Efficiency in Pharma Manufacturing
PAT Can Turn Batch Processes into Continuous Processes
An investment in process analytical technology (PAT) is not just a capital investment, but also an investment in human resources and a commitment to change fundamental processes. PAT is more than just inline analyzing. As an enabler of Quality by Design (QbD), PAT affects all pharmaceutical workflows, from product development to process design to process control.
The most important aspect of a PAT implementation is to retrain a company's culture to approach quality with a proactive rather than reactive mindset. To achieve this, cross-functional, multidisciplinary teams are necessary to effect change management to devise and implement the new processes that take full advantage of PAT. A successful PAT implementation requires a rethinking and a reorganization of resources. Users should anticipate an increased need for statistical analysis and more control engineering skills as analyzers are integrated into production systems. A key success factor, however, is to bring together development and production teams at the earliest stage to ensure the buy-in of top managers of these departments.
On the technical side, it's important to understand that PAT tools and devices won't simply automate existing processes. Instead, processes like manual quality checks are replaced by automated measurements. This information in real time allows the process to be adjusted, but this new flexibility must be built into the process control strategy - a challenge that will require program changes on existing lines, or consume development resources when engineering new lines. In addition, production equipment should have redundant systems in place in the event PAT equipment should fail.
Costs and Risks of Not Investing in PAT
Making a business case for PAT is not straightforward because it is difficult to estimate future returns from improved quality or the lower costs of generating less waste. A PAT investment is not just in equipment, but also in people and processes, so those costs must be amortized over a longer payback period. ARC recommends taking a holistic view to the PAT value proposition that not only considers the initial capital outlay, but also weighs the costs of organizational change against the long-term benefits.
The pharmaceutical industry is known for high-tech research and development but relatively low-tech manufacturing. PAT technology is changing that. In the past, the heavy R&D focus and strict, validated processes made it nearly impossible to improve manufacturing processes with optimization tools commonly used in other industries. Started by the U.S. Food and Drug Administration to proactively improve the productivity of American drugmakers, the PAT initiative has become a chance for all manufacturers to add sophisticated technology to their process control with the cooperation of government authorities, rather than being held back by regulations. As the industry adopts PAT, pharmaceutical manufacturers may not be able to afford not to invest in PAT due to the risk of losing competitiveness.
Making a Batch Process Continuous with PAT
Faced with the pressures of globalization, some pharmaceutical manufacturers are answering their challenges with innovation. One of the top five global pharmaceutical manufacturers recently employed Sipat in a pilot project. Instead of just designing PAT tools into the tablet-making process, the company took the bold step of converting a classic batch process into a continuous process using Sipat. The goal was to take an innovative approach to improve some of the chronic inefficiencies inherent in this type of process, including high inventory requirements, long changeover times, disconnected processes, high process losses and low asset utilization.
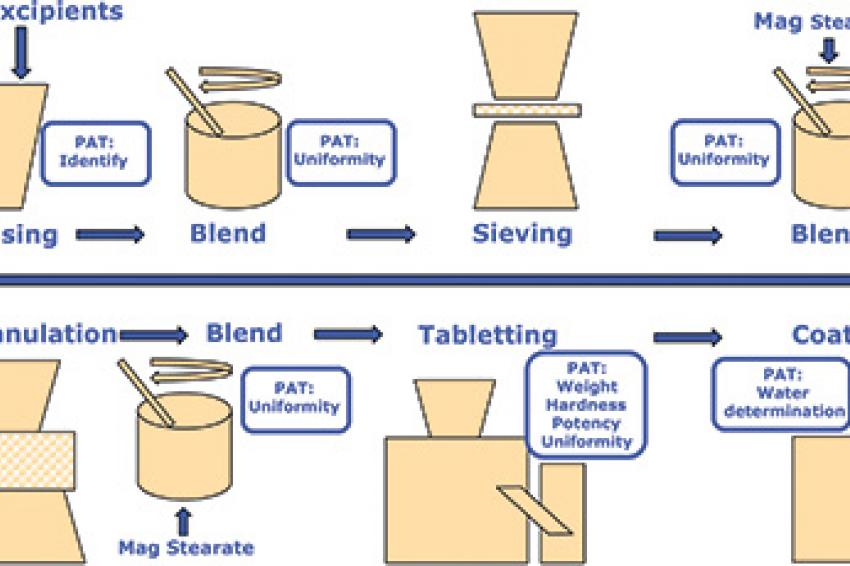
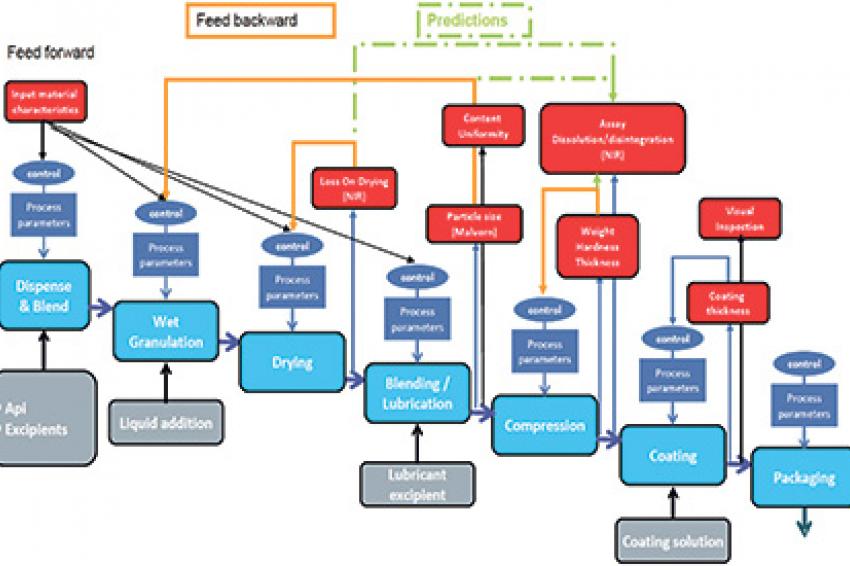
The production line applies high-shear wet granulation process technology to produce oral solid dosages (tablets). The line consists of unit operations for granulation, drying, milling, blending and compression, and is controlled by two PLCs (SIMATIC S7-300) with WinCC for line visualization. Near-infrared (NIR) analyzers measure properties such as moisture, content uniformity and assay, and laser diffraction is employed for particle-size detection.
In this application, Sipat software collects and evaluates multivariate product quality information (CQAs), such as loss on drying or particle-size distribution, as well as univariate process data such as speed, torque, temperature and compression forces for closed-loop model predictive control. Quality parameters are shared with a manufacturing execution system (SIMATIC IT) that tracks this information for real-time release reporting. Sipat allows parameters such as moisture content, content uniformity, hardness and thickness to be adjusted should a batch start to veer out of spec, thus maintaining quality in real time and greatly reducing the risk of having to scrap a whole batch at the end of the process. By using PAT tools, company engineers were able to deepen their process understanding much faster. The scale of the process equipment used for continuous manufacturing in the development phase is the same as for commercial production, so the company was able to eliminate the scaling-up process.
According to the manufacturer, the new continuous process concept was developed within two weeks and the line was producing tablets reliably and with excellent quality after just six months. The in-line quality checks now make possible real-time release with significant increase in efficiency versus traditional batch methods with end-of-line QC. Interestingly, the equipment has a substantially smaller footprint - requiring only about a tenth of the space needed by an equivalent batch line. The reason is that a batch process requires all raw materials to be stored locally during the batch process. A continuous process, on the other hand, employs a continuous flow of materials that don't need to be stored at a single location. The result is smaller requirements for clean room space, lower clean room HVAC energy usage, fewer operators, and less work-in-process.
Hot Melt Extrusion as a Continuous Process at MSD
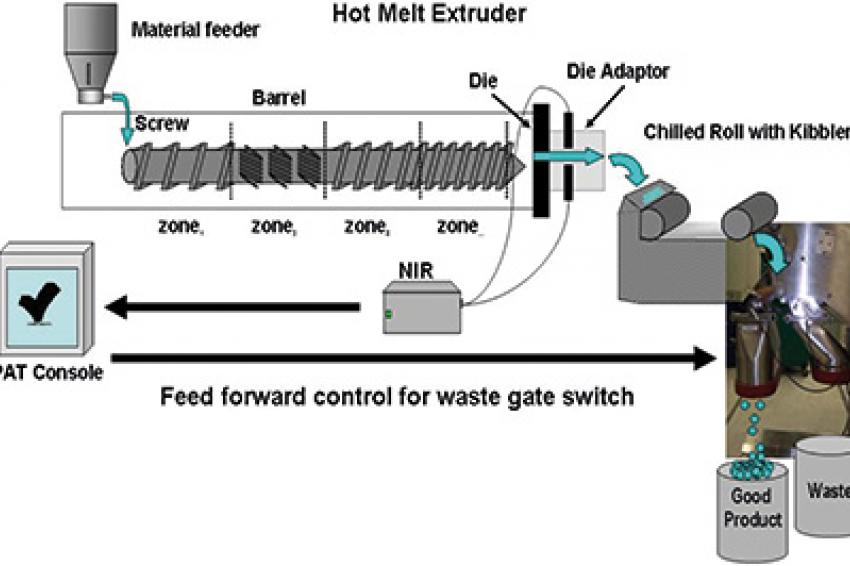
Extrusion is a process commonly used to manufacture plastic parts by forcing molten material through a die under pressure. Merck Sharp & Dohme Corp. (MSD), a subsidiary of Merck & Co., employs a hot-melt extruder (HME) in a process for manufacturing solid oral dosages. This process allows a drug substance to be stabilized in its amorphous form within a polymer matrix, increasing solubility and in vivo exposure, which is useful when the active pharmaceutical ingredient has low solubility. The HME process combines multiple steps such as mixing, melting, degassing and densification, which increases efficiency by reducing the number of unit operations. MSD employs PAT tools and Sipat software from Siemens, using Fourier-transform near-infrared (FT-NIR) spectroscopy to monitor form change, assay and uniformity of the mixed substance.
Extrusion is essentially a mixing process during which process parameters for temperature, mixing and feed rate can be adjusted. Extrusion occurs when the mixture is forced under pressure through an orifice in the die. As the extrudate leaves the die, the molten substance drops between two chilled rolls, causing its temperature to fall below its glass-transition temperature and form a brittle glass sheet. The sheet is then broken into smaller flakes of glass to make it easier to mill. In the final steps, the material is milled, blended with excipient, and then lubricated and compressed into a conventional solid oral dosage form.
The extruder's screw and barrel are modular, allowing flexible control of venting, material feed, mixing, heating and cooling. PAT tools (NIR, Raman, ultrasound or UV-VIS spectroscopy) come into play at the moment of extrusion. These devices interface to Sipat software from Siemens, allowing CQA deviations (measured here as percent drug loading [wt/wt]) to be identified in real time. If drug-loading predictions are outside of specification, alarms trigger, immediately activating a switch gate to divert out-of-spec material to waste. Any CQA deviations are brought back into line by adjusting the necessary process parameters.
According to MSD, the HME platform can be easily scaled up to production levels or scale-up can be eliminated, depending upon product volume forecasts, because the extruder used for most of process development is essentially the same size as the one used later in production. If scale-up is required, CQAs remain the same while process parameters are scaled up to manufacture large batches of extrudate - up to several thousand kilograms. After the successful development and scale-up of a pilot project, MSD has now deployed the HME platform to additional sites.
Last Word
Quality by Design takes a different approach to ensure consistent levels of quality by allowing for flexibility during the manufacturing process. PAT tools play a crucial role in the implementation of QbD by providing the real-time feedback that allows operators to keep CQAs in spec by adjusting processes during operation. At the same time, quality data collected during the process can take the place of end-of-line QC, drastically cutting batch release times. The many business benefits of employing PAT tools are highly attractive.
This is the second of two articles from David Humphrey on process analytical technology (PAT). Part 1 was published in issue 3-4/2015 of CHEManager International.
Contact
ARC Advisory Group GmbH & Co KG
Stadttor 1
40219 Düsseldorf
Germany
+49 2104 542 012